Vyvgart Hytrulo (efgartigimod alfa and hyaluronidase-qvfc) for myasthenia gravis
What is Vyvgart Hytrulo for myasthenia gravis?
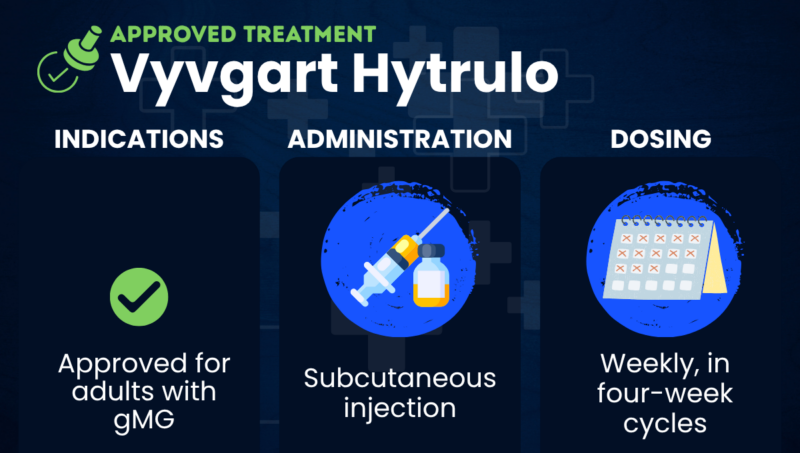
Vyvgart Hytrulo (efgartigimod alfa and hyaluronidase-qvfc) is an approved injection therapy used to treat adults with generalized myasthenia gravis (gMG), regardless of whether they have known MG-causing antibodies.
In myasthenia gravis (MG), self-reactive antibodies target proteins needed for nerve-muscle communication, disrupting the process and causing muscle weakness. These antibodies belong to a class called immunoglobulin G (IgG). Vyvgart Hytrulo works by blocking the neonatal Fc receptor (FcRn), a protein that normally helps stabilize IgG antibodies and prevent their destruction. Blocking FcRn helps increase the breakdown of IgG antibodies, including disease-driving antibodies, which lowers their levels and can ease disease symptoms.
The medication, sold by Argenx, also contains a version of the hyaluronidase PH20 enzyme, which enables it to be delivered via under-the-skin, or subcutaneous, injections. This may offer more convenience than Vyvgart, the company’s older formulation of efgartigimod alfa that is given by a healthcare provider via infusions into the bloodstream, or intravenously.
In May 2026, the U.S. label for Vyvgart Hytrulo and Vyvgart was expanded to cover adults with gMG regardless of antibody status. Vyvgart Hytrulo is also used to treat the rare immune-mediated condition chronic inflammatory demyelinating polyneuropathy.
Therapy snapshot
| Brand name | Vyvgart Hytrulo |
| Chemical name | Efgartigimod alfa and hyaluronidase-qvfc |
| Usage | Used to treat adults with generalized myasthenia gravis |
| Administration | Subcutaneous injection |
Who with myasthenia gravis can take Vyvgart Hytrulo?
Vyvgart Hytrulo is approved in the U.S. for adults with gMG. The medication is sold in the European Union and Canada under the name Vyvgart SC for adults with gMG who are positive for antibodies against the acetylcholine receptor (AChR), the most common type of MG-causing antibody.
Vyvgart Hytrulo is contraindicated, or should not be used, in people who have had serious immune reactions, called hypersensitivity reactions, to efgartigimod alfa, hyaluronidase, or any of the other components in the therapy.
How is Vyvgart Hytrulo administered in myasthenia gravis?
Vyvgart Hytrulo is given via subcutaneous injection into the abdomen. The prefilled syringe may be administered by patients or caregivers after proper training, while the vial formulation is administered by a healthcare professional.
For people with gMG, Vyvgart Hytrulo is initially given once weekly for four weeks. The decision to start subsequent four-week treatment cycles depends on a person’s clinical response.
The prefilled, single-dose syringes contain 1,000 mg of efgartigimod alfa and 10,000 units of hyaluronidase, and are administered over 20 to 30 seconds. When administered by a healthcare provider, the medication may be supplied in single-dose vials containing 1,008 mg efgartigimod alfa and 11,200 units of hyaluronidase. Injections from the vial take 30 to 90 seconds.
Vyvgart Hytrulo in myasthenia gravis clinical trials
Vyvgart Hytrulo’s approvals were supported by three Phase 3 trials. Two of these studies demonstrated the benefits of intravenous Vyvgart in adults with gMG, while the other showed that Vyvgart Hytrulo was expected to have comparable effects to the original Vyvgart formulation.
- The Phase 3 ADAPT trial (NCT03669588) demonstrated the benefits of Vyvgart in adults with gMG who were positive for anti-AChR antibodies. The data showed that significantly more people on Vyvgart than on a placebo experienced meaningful reductions in patient- and clinician-rated measures of disease severity.
- The Phase 3 ADAPT SERON study (NCT06298552) supported the expanded use of Vyvgart in adults with gMG who were AChR antibody-negative, including individuals who tested positive for other MG-causing antibody types or negative for any known disease-related antibody. The data showed that Vyvgart led to meaningful reductions in disease severity in AChR antibody-negative patients.
- The Phase 3 ADAPT-SC clinical trial (NCT04735432) demonstrated that Vyvgart Hytrulo was at least as effective as Vyvgart in its ability to lower IgG levels in adults with gMG. The subcutaneous medication also led to reductions in disease severity similar to those seen with Vyvgart.
Vyvgart Hytrulo side effects
The most common side effects associated with Vyvgart Hytrulo include:
- respiratory and urinary tract infections
- headache
- injection-site reactions
If injection-related reactions occur, a doctor should consider the risks and benefits of giving Vyvgart Hytrulo again based on the severity of the reaction. In some cases, medications to prevent a reaction may be recommended before subsequent injections.
Vyvgart Hytrulo also carries warnings for other potentially serious side effects that require careful monitoring. These include:
- infections
- hypersensitivity reactions, including anaphylaxis, a life-threatening systemic reaction
If any of these occur, Vyvgart Hytrulo may need to be paused or discontinued, depending on the severity of the reaction.
Vyvgart Hytrulo treatment should be delayed in a person with an active infection. Before starting a new treatment cycle, doctors should review whether patients need any age-appropriate vaccines recommended under immunization guidelines. Live vaccines are not recommended during treatment.
Myasthenia Gravis News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by