Myasthenia Gravis News Community › Forums › Research and Clinical Trials › What happened to efgartigimod/Vyvgart for seronegative MG?
-
What happened to efgartigimod/Vyvgart for seronegative MG?
Posted by MG Community Member on December 24, 2021 at 4:29 amAs of October 2021, there were reports from the phase III ADAPT study that efgartigimod/Vyvgart was found to be effective for seronegative MG. Then on December 17, 2021, the FDA approved this medication only for AChR-positive patients. What happened?
I have seronegative generalized MG and am currently taking prednisone, loads of pyridostigmine/Mestinon, and methotrexate. I took Cellcept (10 months) and Imuran (22.5 months) but had to discontinue both due to side effects. For the past three years, in order to be somewhat functional, I have had to undergo routine plasma exchange. I am grateful that my insurance pays for the plex, and I have managed to stretch it to every two weeks, but I would prefer not to have to do it. Therefore, my neurologist and I were looking forward to FDA approval (and insurance coverage) for efgartigimod. Does anyone know if Argenx is planning to submit efgartigimod/Vyvgart for FDA approval for seronegative MG?
-Nan
MG Community Member replied 1 year, 11 months ago 21 Members · 47 Replies -
47 Replies
-

MG Community Member
MemberDecember 24, 2021 at 11:03 pmMe too. My neurologist has been looking forward to the approval for my treatment. I had read the previous press releases and it was submitted for all gMG patients. I’m seronegative and have had serious side effects from prednisone, imuran, cellcept, tacrolimus, IVIg, PLEX, and most recently, Rituxan. I’m out of options. Argenx website celebrates its approval for the most common form of gMG. What about the rest of us?
-

MG Community Member
MemberMarch 10, 2022 at 8:56 pmToday, March 10, 2022 I received an infusion of Vyvgart without any issues so far. I will get 3 more infusions one week apart. I still have the catheter for Plex in case the Vyvgart does not work. As far as I know the medication is FDA approved and supposedly Medicare approved. I have MG in the facial area for 4 years. Went through many Plex infusion which did help me immediately. However when I get the symptoms they arrive within days and I can not swallow food, water and talk little. My tongue gets lethargic and go quiet. During a flair up the mucus that suddenly accumulates in my mouth chokes me so I am careful drinking since all muscles in the mouth and throat fatigue fast. I had my thymus gland removed 6 years ago prior to any symptoms of MG.
-

MG Community Member
MemberMarch 10, 2022 at 9:05 pmJoe,
I am guessing you are not seronegative, correct?
I hope you get the remarkable positive results they tout.
I’ve heard first or second infusion gets positive results.
keep us appraised please.
Good Luck.
-

MG Community Member
MemberSeptember 8, 2023 at 3:21 pmThis may sound a bit depressing but vyvgart was not the help I had hoped for. It worked for a month and I thought a miracle happened. And then it worked less. And less. Stopped working completely on my eyes. Helped a few days a week. Then you are supposed to stop for like 3-4 weeks. Forget it. I ended up in the hospital doing plasma exchange. Ugh. I’ve yet to find anything that will last and help with the swallowing and speech. The prednisone still works best with the eyes.
-

MG Community Member
MemberMay 21, 2024 at 1:15 pmSorry to hear your problem.
-
-
-

MG Community Member
MemberDecember 25, 2021 at 12:38 amI too was looking forward to the approval and my neuro thought it would get approved for us as well.
i meet with him on 1/10 hoping to get info.
I started IVIG 6 weeks ago hoping it to be a short term deal.I wonder if the drug company felt the approval would get the best chance of approval this way and we seronegative people will have to get approval off label which is how IVIG is anyway.
we will see……merry Christmas to all.
-

MG Community Member
MemberJune 18, 2022 at 9:25 pmI met with my Neuro beginning of June, she is hoping as am that the drug gets approved soon for us seronegative people. So far, one year of Cellcept, 90mg Pred, weaning to 20mg now, gone through Ivig, Plasmapherisis, Mestinon is hard to tolerate and doesn’t really work for me any more. My drs are going to have a big POW WOW to discuss next course of treatment. Meanwhile, Ptosis worse, muscle weakness worse, and hyperventilating.
-
-

MG Community Member
MemberDecember 25, 2021 at 12:57 amDoesn’t the drug work by clearing the auto antibodies to acetylcholine receptor. The obvious approval are acetylcholine positive. Then they have to study if it works for other types. Doctors can still use it off label just have to get approval from insurance.
-

MG Community Member
MemberJanuary 1, 2022 at 2:47 amThey actually included I think 27 people or so in the study and they had mostly positive results. But I agree they likely have to have an independent study with just seronegative. Hopefully we won’t be neglected as there not nearly as big a group of us. That’s why I wonder if they will just use it off label for us.
time will tell.-

MG Community Member
MemberJanuary 4, 2022 at 12:06 amTheresa and Jennie, it looks like we are all in the same boat. The problem with Vyvgart being prescribed off label, is that our health insurance almost certainly won’t pay for it. Especially if it is expensive, which I suspect it is.
Theresa, if you don’t mind, please post (or PM me) and let us know what your neurologist says at your January 10th appointment. My next neuro appointment is January 20th.
-

MG Community Member
MemberFebruary 11, 2022 at 7:21 pmNan,
I have the option to go on vyvgart whenever I’m ready.
i speak to neuro again Monday.
I figure nothing to lose but 4 weeks.
im on Medicare and a BCBS supplement and he is confident he can push it through . It is Barrow’s institute so they have the systems and staff to make this sort of thing happen. I’ve had many occasions over the years where one doctors office could make a treatment happen when another could not. Not necessarily MG. Even getting IVIG is off label for seronegative.
so it can be done.
-
-

MG Community Member
MemberMarch 1, 2022 at 3:02 amThe mechanism of action is to block the FcRn (neonatal (formally fetal) receptor) which is needed to create IgG antibodies— almost all auto immune diseases result from production of IgG antibodies which erroneously target and interfere with the normal function of a receptor (such as the ACh receptor in MG). There is no reason to suggest it would not work in patients who are seronegative in whom PLEX works. The data submitted to the FDA showed a trend of improvement in the small number of seronegative patients included; however, statistical significance was not achieved. I’m not sure how to go about it, but we must petition the FDA to reconsider approving the drug as all current clinical studies have excluded seronegative patients; alternatively we could petition the drug company to perform a clinical trial in seronegative patients, but that is not cost-effective for them; therefore unlikely.
-

MG Community Member
MemberMarch 1, 2022 at 3:06 amI’m not sure how to go about it, but we must petition the FDA to reconsider approving the drug as all current clinical studies have excluded seronegative patients; alternatively we could petition the drug company to perform a clinical trial in seronegative patients, but that is not cost-effective for them; therefore unlikely.
-

MG Community Member
MemberMarch 9, 2022 at 7:48 pmOr perhaps a class action law suit.
many attorneys out there?may have to do each insurance company starting with Medicare for off label use. ?
-
-
-

MG Community Member
MemberJanuary 21, 2022 at 11:04 pmI had my neuro appointment yesterday. My oral methotrexate dosage has been increased to 15mg. per week. Since he ordered bloodwork to check my immunoglobulin levels, I guess he is considering IVIG then subcutaneous IG if my antecubital veins continue being recalcitrant. Fingers crossed 🙂
-

MG Community Member
MemberFebruary 11, 2022 at 8:15 pmHi
I would really like to reach out to this group to find out what is known about the non-responders to efgartigimod and nipocalimab.
In phase 3 efgartigimod works for 68% (great news) but 32% it does not in gMG
Likewise, in phase 2 nipocalimab works for 52% and 48% it does not.On non-responders is their a high baseline of IgG or have neutralising antibodies to the therapy started? Or something else?
Any discussion greatly appreciated
-

MG Community Member
MemberMarch 9, 2022 at 9:08 pmHi Folks –
I wish I could get some Vyvgart!!
Kaiser will be doing its own pharmacy evaluation.
Best wishes, Wayne
-

MG Community Member
MemberMarch 10, 2022 at 2:30 pmI keep hoping they announce an extended study for seronegative patients!
-

MG Community Member
MemberApril 5, 2022 at 3:39 pmAnyone know if you have to undergo other treatments prior to starting Vygart to get it approved. I had to go off the imuran when I had COVID then was forced by my workplace to get the booster 3 wks after I had it. I can’t get back onto it. Everytime I restart, I get terrible vomiting without warning. Not really pleasant when a bathroom is far away and I work in healthcare. Do you need to try plasma or IVIG before they will approve Vygart?
-

MG Community Member
MemberApril 5, 2022 at 3:40 pmOh, also, I am antibody positive
-

MG Community Member
MemberApril 5, 2022 at 4:34 pmSheila
I’m sure each insurance has its own protocol.
I would ask your doctor to prescribe it and see if you get coverage. Also contact Vyvgart yourself and ask how to get it. You can log into their site and it will tell you who to call. There is also grants through them to cover copay up to $25,000. I believe.
you are antibody positive so you qualify.
seronegative do not. I start in 10 days it took some work and not accepting the first denial to get it approved.
I got lucky.
Vyvgart definitely wants to get it to everyone and are very happy to help.-

MG Community Member
MemberOctober 16, 2023 at 5:53 pmThe neuro tested and it was at first equivocal. He retested and seronegative. Had other antibody tests musk etc also negative. Really bummed out. Limits your options if your abt negative. Would like to know when by Gary might be approved for seronegative
-
-

MG Community Member
MemberMay 5, 2022 at 3:45 pmHere’s the saving grace for us seronegative folks, Imuran, Cellcept, Retuximab, IVIG and many other treatments are not FDA approved for MG and most are covered for use with most insurance companies.
-

MG Community Member
MemberMay 12, 2022 at 10:14 pmI completed my 4th infusion of vyvgart last week.
about half way into the 2nd infusion, I began noticing subtle things. Less naps, forgetting mestinon.
Then 3rd and 4th infusion, those and other “subtle” changes.
after the 4th, the next day was a big improvement. Not like ready for a marathon, but gradual daily improvements. Much like the snowflake description of our disease. Not every day the same but of course the more I do, a couple days later I might get pretty tired. Much different though.
note when you have had the disease for 10 years, we naturally schedule our time so this habits don’t disappear immediately.
I am convinced there is a significant improvement. If it had to out a number to it, I would say I am 70-75% better, maybe a little more. Hardly any SOB, few and shorter naps.
I can go all day, sometimes till 11 pm.I’ve been told that it can take 2 or even 3 cycles before a patient might experience improvement. But it is real.
I hope this becomes available to all Seronegative patients. It’s worth the opportunity.‘absolutely no side effects.
down to 7.5 mg prednisone.
i have to remember that reduction will cause MG like symptoms.
good luck to all.
-

MG Community Member
MemberMay 13, 2022 at 1:31 pmHello Theresa, That’s fantastic news, glad the therapy seems to be working for you. Can you clarify “i have to remember that reduction will cause MG like symptoms.”? Are you saying you become symptomatic because of the taper, but it’s not the MG flaring? I’ve been in IG for a couple years. I’m down to 7mg a day, but am afraid to go further. Every time I taper, I have droopy eye symptoms, but a couple weeks later everything clears up.
Mike
-

MG Community Member
MemberMay 13, 2022 at 6:39 pmSorry, reduction of prednisone, causes bothe it’s own side effects, but also can cause flaringnof the autoimmune disease for which you are taking.
so yes, my eyelid has been real droopy with .5 reduction in spite of the VyVgart benefits.
I want off the prednisone so I have to keep that in my mind that it is quite possibly the steroid reduction and that the treatment is all good.
and overall it is.
daily I can see my activity level, fatigue, SOB symptoms are all better since that last infusion.
I am Seronegative so super excited it is working.hope that clears up my explanation.
-

MG Community Member
MemberMay 13, 2022 at 6:43 pmMike just reread your comment.
i was reminded yesterday by a veteran MGer, she said to remember our eyelids have the smallest of nerves to control them and they are the most outlying. So it is natural they would respond Accordingly.
I have to jow drop .25-.5 mg every 3-4 weeks so my symptoms are not too profound. I find after 3-4 weeks they subside.
im at 7.5 mg right now. Down from 20 beginning in November.
it’s a slow haul. I have tons of side effects from them so want them gone. Just praying the VyVgart gets me there.
good luck!
-

MG Community Member
MemberMay 16, 2022 at 2:04 pmOkay…I got it. Just like me. I’ve been living with MG for 26 years. I can only go ,5mg every 2 months. I’ve been on Prednisone the entire time and have so many side effects. Slow wins the race. I’ve been on IVIG for 2.5 years. Any therapy takes a very long time to respond with me, so don’t get discouraged. It’s taken me this long to get from 22.5mg per day down to 7mg per day. Be patient….don’t let a time table dictate your tapers….let the MG symptoms do the dictating.
-
-
-

MG Community Member
MemberMay 17, 2022 at 1:35 pmThis is why there is no FDA approval for us seronegative ab patients.
https://www.argenx.com/news/argenx-announces-positive-topline-phase-3-adapt-trial-results
The trial was done on both AChR positive and negative patients. There are 2 reasons the FDA did not approve the drug for seronegative and musk patients, is because the sample was too small and
Percentage of efgartigimod responders on the MG-ADL score in the AChR-antibody negative patient population was consistent with the AChR-Ab+ patient population, but a greater placebo response was observed in this cohort.
-

MG Community Member
MemberDecember 29, 2022 at 4:01 amLike Theresa, I am seronegative. Finished my 4th Vyvgart infusion last week and I feel SO MUCH better. I pray it will stick. I forget to take mestinon now as my body isn’t reminding me to take it, like it used to with the constant muscle weakness. Still having difficulty with swallowing food but my legs are stronger and I’m walking better,
-

MG Community Member
MemberAugust 10, 2023 at 2:11 pmIn the quaint town of Eldenwood, nestled among rolling hills and ancient forests, lived a young girl named Elara. She possessed a rare and mysterious condition known as “Aeris Veil,” a disease that caused her lungs to be unusually sensitive to the slightest changes in air quality. Her life was a delicate dance between fragility and strength.
-

MG Community Member
MemberSeptember 7, 2023 at 8:56 amI am Seronegative. (3 different neg ACHR tests, 2 from Mayo). Diagnosed 2.5 years ago. Sudden onset, double vision, eye droop, leg weakness/fatigue, swallowing issues. Hospitalized 4 times. For 2 years been on Cellcept, Mestinon 4x, and Prednisone (now down from 60 to 13.5). Eye issues gone but severe leg weakness/fatigue remain. Have swallowing issue where late in day get “wad” in throat…feels like have to swallow but can’t. (but no choking or chewing issues…yet) Have had 2 singled fiber emg’s that were “abnormal” and showed “jitter” to confirm diagnosis. Am treated at a major University Medical Center.
I yo-yo. Mostly down all day. Good periods can fix a sandwich. Bad times can’t stand long enough to fix bowl of cereal or barely walk to table. Heat -even a tiny little – is an instant trigger, as is walking for 4-5 minutes. Any trigger and weakness/fatigue/swallowing sensation gets worse. Can exercise some in pool and do that 3x week except when setbacks. But have to go to place were is just few steps from drop off entrance to pool, and have to use wheel chair to go into doc or hospital. Can’t walk in a store. Before onset was biking every morning and walking 2 mi 3x per week and physically very active. I could push through pain and fatigue if didn’t make me worse. So mostly housebound for 2 years.
Had IVIG every 2 weeks for year and a half. I was better then, could do a little more, but out of blue caused blood clots. (All genetic tests said I was neg for any genetic propensity to clotting). So had to discontinue and got worse. Mostly bad now.
One year ago found pressure on spinal in neck and surgery by a top surgeon went well. My neuro MG doc wants to blame symptoms on that. Neuro surgeon says after 2 MRI’s and Myleogram, no, not the problem now
MG doc says I am not bad enough for Vygart now because my ADL scores bounce from 1-4. And my “sitting strength tests” are good. But that is only because much of that test measures eye and chewing problems which I admit I don’t have. And only because I am mostly chairbound conserving energy. If I tried to live any decent physical life could easily put myself in hospital. But seems to me I have all the MG signs…extreme susceptibility to heat, fatigue with very little exertion, legs weigh 30 lbs each, overdoing or heat exposure brings strong delayed reactions which take days/weeks to overcome. Swallowing is only a problem after overdoing it and getting weak.
Also, recently diagnosed with mild RA Ankylosis Spondylitis. Now MG doc attributing problem to that. Have 2<sup>nd</sup> appointment with Rheumatologist, since recent MRI RA AS diagnosis, to discuss plan forward. Good thing is, the two docs are willing talking.
Questions –
1. Am I imagining how severe my MG is?
2. Has anyone with lower ADL scores still been put on Vygart? What is experience with Medicare approving Vygart or does that have to go to your supplemental ins?
3. Is anyone being treated for both MG and RA. What is regimen?
-

MG Community Member
MemberSeptember 8, 2023 at 7:05 pmNan and Theresa – Thank you for your very encouraging remarks about Vyvgart! I have Kaiser in Northern California. I’m about to change neurologists because my current neurologist has resisted my desire for Vyvgart.
I’m on 40 mg of prednisone daily. I make smoothies loaded with collagen and other proteins and tomorrow I get my 2nd bone density scan in a couple years, so I can compare. I also have a continuous glucose monitor to track my blood sugar all day long.
I understand that there is no contraindications regarding Vyvgart and prednisone..
I’ll keep you posted.
Thanks again!
-

MG Community Member
MemberSeptember 8, 2023 at 10:37 pmWayne, best of luck to you. Keep us posted!
-
-

MG Community Member
MemberSeptember 8, 2023 at 7:08 pmMy bone density scan showed a significant improvement in density… pretty remarkable! Collagen supplementation is a must when taking prednisone. I recommend it for all humans, in fact! 😁
-

MG Community Member
MemberSeptember 8, 2023 at 7:13 pmFYI: The post prior to my last post post was a re-post of other chat, so the date is wrong.
-

MG Community Member
MemberSeptember 8, 2023 at 11:01 pmI am seronegative with a positive SFEMG and a lung function test with very low MIP and MEP which my neuro used to get Medicare approval for Vyvgart. It has been a miracle drug for me. My score before my first infusion was 12. By the end of the first cycle, it was 2. I am now doing a second cycle. It took care of most of my symptoms but I still had fatigue.
-

MG Community Member
MemberSeptember 9, 2023 at 5:23 amThat is great news! So happy for you. By score, were you referring to the ADL score?
Cheers
-

MG Community Member
MemberOctober 25, 2023 at 1:56 pmYes, and a month out from my second cycle and I’m hanging in there with a 4!
-
-
-

MG Community Member
MemberOctober 31, 2023 at 8:44 amBuckeye, I too have Seronegative MG, Ankylosing Spondylitis in my lumbar region and Spondylolisthesis in the cervical area of my spine. It was diagnosed per MRI after I had severe neck pain, headaches, eye pain and and numbness in my arm, along with pain running down my leg, and thigh. My rheumatologist recommended steroid injections in both places which have helped somewhat.
MG is being treated with IVIG, 120mg of Mestinon daily and occasional prednisone.
IVIG helped more at first but symptoms are increasing again, including increased shortness of breath. I’m meeting with my neuro’s nurse practitioner to discuss it. At 72yo I’m not eligible to have my thymus removed.
Many of the symptoms I have are related to all of the conditions I’ve been diagnosed with. It can be very confusing. Since the symptoms are progressing, I need to figure out what’s causing them and with my docs, figure out a treatment that’s more effective. My sed rate has been very high at 120 for several months now. My neuro has said it could be caused by IVIG. I’m now in the process of ruling out what else could be causing the rapid increase in inflammation. Hate to eliminate IVIG until something else can replace it.
So that’s where I am now. Thanks for sharing your insights on what’s happening with you. Please continue to share. It’s helpful in so many ways.
-

MG Community Member
MemberNovember 5, 2023 at 8:15 amI started on Cosentyx 4 weeks ago for the RA ankylosing spondylitis. The day I took the first shot my double vision came back big time. First time in 2 years that I had had it.Two separate planes when I looked up, returned together in phase when I looked straight ahead. Have had 4 weeks of loading shots now, and vision is still a problem. MG doc and RA doc both say there is no evidence connected…. But nothing in two years and now back? Next Cosentyx shot is month away so we will see…
Haven’t seen any effect of Cosentyx yet on any pain or weakness… Have extreme hip pain from RA which may be aggravating MG. My doc has a clinical trial going with Vygart on Seronegative patients but won’t put me in it because “I have too many issues!” I don’t think I would have so many issues if could get my MG muscle fatigue under control…
-

MG Community Member
MemberNovember 5, 2023 at 8:35 amYour RA is autoimmune so in my head a drug designed for that could be good for MG. I have to wonder if this is how they learn to use certain drugs off label .
I am sure vyvgart, which I take, and am seronegative , and so many others are being developed for many autoimmune diseases. Rituxin originally was for lupus or Ra?, and is used for many off label.
Keep reporting your findings, possibly to the manufacturer if it continues.
With vyvgart I’ve had 16 days one 10 one 6 Of total remission eyes included. Usually my eyes are unaffected. This infusion Monday my eyes are 70% good. Sure makes it hard to keep eyeglasses up to date. 🤦♀️
-
-

MG Community Member
MemberNovember 5, 2023 at 8:41 amJoanne
I too initially had positive effects from IVIG, then 3 months later it just made me sick. I quit and went on Vyvgart.
I have Medicare AB and Humana pard D who approved the vyvgart, then what they don’t cover is done so by The Assistance Fund. Check and see if there are still available positions there. It is a godsend both vyvgart and the assistance fund.
I’m 70 and at this stage don’t have time to play around with treatments that don’t work well. I also still take 7 mg prednisone and 300 mg of mestinon a day.
Talk to your neurologist and see if he can get you something better than IVIG.
-
-

MG Community Member
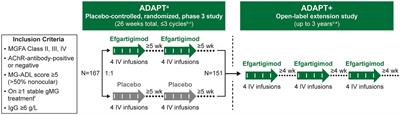
MemberJanuary 31, 2024 at 3:48 pmHas anyone seen the results from the new Adapt + Vyvargt study? Looks like this could be good news for Seronegative patients. Anymore Seronngative folks getting Vyvargt and is it helping? Thoughts on if this new study will help get insurance to approve it?
https://www.frontiersin.org/articles/10.3389/fneur.2023.1284444/full
frontiersin.org
ObjectiveADAPT+ assessed the long-term safety, tolerability, and efficacy of efgartigimod in adult participants with generalized myasthenia gravis (gMG).MethodsADAPT+ was an open-label, single-arm, multicenter, up to 3-year extension of the pivotal phase 3 ADAPT study. Efgartigimod was administered in treatment cycles … Continue reading
-

MG Community Member
MemberMarch 8, 2024 at 2:24 pmSeronegative and just got approved for Vyvgart. Start my first infusion Monday Mar 11-24, at the hospital. Once a week for 4 weeks and then evaluate going to once a month doctor said.
Insurance approved but don’t know my cost yet.
We will see what it does. Also have ankylosing spondylitis andon Cosentyx for that so will see how two together do.
Will report back.
-

MG Community Member
MemberApril 23, 2024 at 2:35 pm💃🏽 Yay!!!
Log in to reply.