Robot-assisted Thymectomy Considered Safe for MG Patients, Singapore Study Says
Written by |
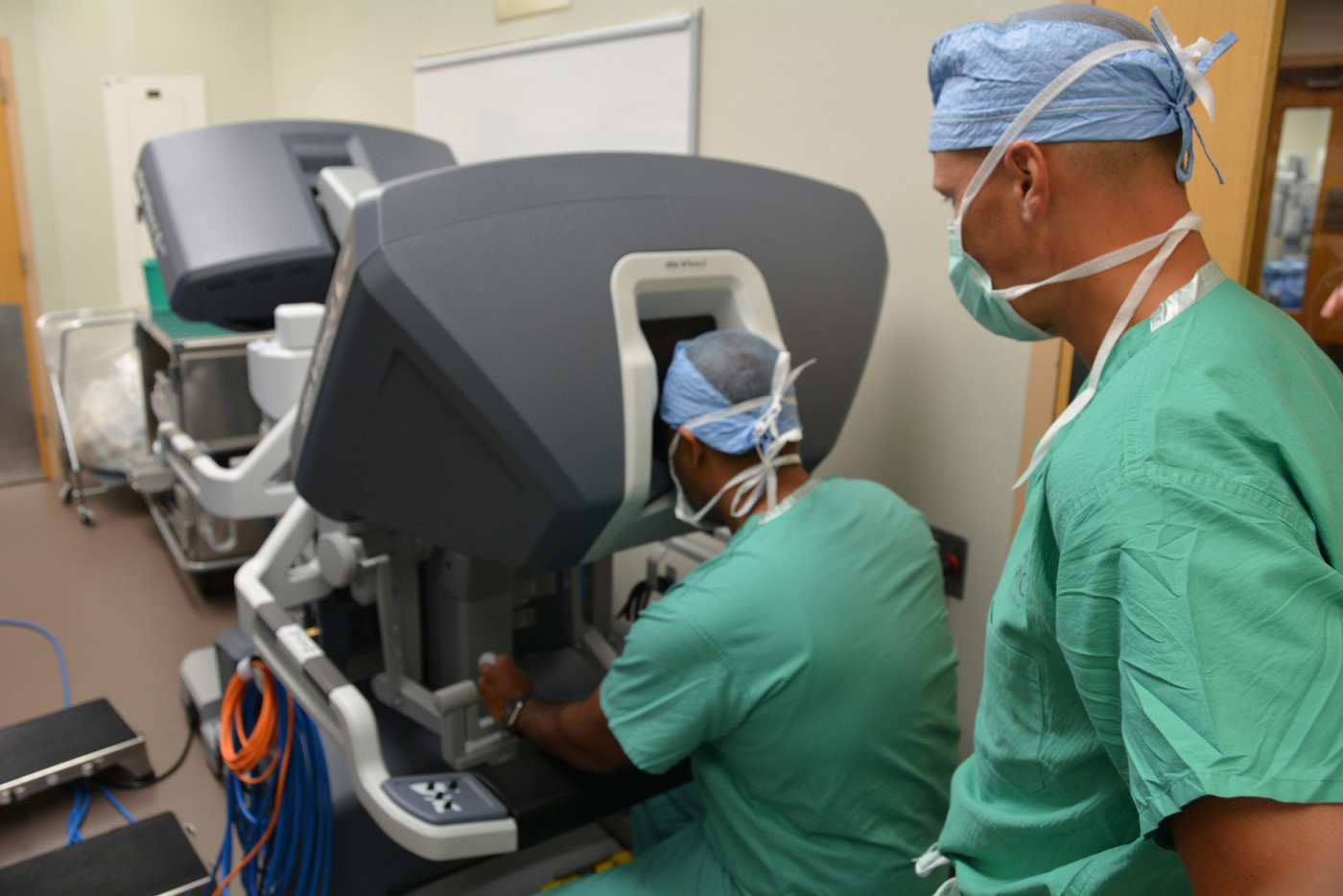
Robot-assisted surgery to remove the thymus gland is considered a safe method of performing a thymectomy on myasthenia gravis patients, a recent study in Singapore shows.
The study, “Robotic Thymectomy in Patients With Myasthenia Gravis,” was published in the journal Heart, Lung and Circulation.
Thymectomy is one of the major treatment options for myasthenia gravis. It is a surgical procedure to remove the thymus gland, a small gland under the breastbone that is important for the production and maturation of immune cells.
Although the origin of myasthenia gravis is unknown, researchers believe the thymus gland plays an important role in producing autoantibodies that mistakenly attack the muscle-nerve connections in patients.
A recent Phase 3 clinical trial (NCT00294658) showed that thymectomy significantly improved the symptoms of myasthenia gravis in patients younger than 65 who were on prednisone and did not have thymoma, a thymus tumor that is either benign or malignant.
Patients with mild symptoms also can become candidates for surgery if they have respiratory or swallowing difficulties.
The study’s goal was to evaluate the safety and clinical outcomes of patients who had their thymus removed via robotic thymectomy, a recent surgical technique in which a surgeon is assisted by a pair of robotic arms coupled with a small video camera inserted into a patient’s chest.
The U.S. Food and Drug Administration approved two robotically-assisted surgical devices: the Da Vinci Surgical System and the Senhance Surgical Robotic System.
In this study, a team of researchers at Tan Tock Seng Hospital in Singapore retrospectively analyzed the clinical outcomes of 21 patients who underwent robotic thymectomy at the hospital from 2012 to 2015, including 10 men and 11 women. Their mean age was 47.
Patients had very mild to moderate symptoms, according to MGFA classification. Surgeries generally lasted about 100 minutes. One patient had to switch to a different surgical technique, called median sternotomy (a vertical incision along the breastbone), due to the risk of compromising large blood vessels.
The patients’ median length of stay in the hospital was two days. Seven patients required routine follow-up in intensive care in days following surgery.
Biopsies of the removed thymuses confirmed that 13 of 21 patients had a tumor. No complications or early mortality were observed following the surgeries.
“Our results suggest that robotic thymectomy can be performed safely for patients with myasthenia gravis,” researchers stated, adding that “further studies are required to assess long-term outcomes.”


Leave a comment
Fill in the required fields to post. Your email address will not be published.